Collision with a Mars-size body — the same body thought to have formed the Moon — might also have brought life-supporting volatile elements to Earth.

Don Davis / The New Solar System
Scientists have long hypothesized about the origins of Earth’s volatiles. These low-boiling-point elements, such as carbon, nitrogen, and sulfur, are necessary for life, but they weren’t present at Earth’s beginnings. Now, researchers at Rice University propose that volatiles could have arrived via a single collision with a Mars-sized planetary object — the same event that created the Moon. Their findings are published in the January 23rd issue of Science Advances.
Core vs. Crust
When Earth started forming about 4.5 billion years ago, it accreted gas, dust, and debris from its surroundings into a molten soup. Any volatiles would have boiled out of this hot, early mixture. Nevertheless, the existence of life suggests that something delivered volatiles later on.
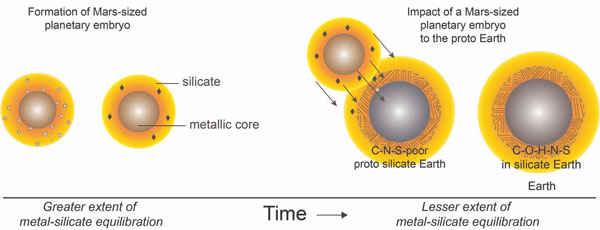
Scientists have long proposed that pristine carbonaceous chondrites — meteorites rich in volatiles — flew in from the outer part of the solar system and collided with Earth in the very late stages of our planet’s formation. The problem with this explanation, says the paper’s lead author, grad student Damanveer Grewal, is that the carbon to nitrogen ratio in carbonaceous chondrites is 20:1. But the bulk of Earth’s silicates (its mantle, crust, oceans, and atmosphere combined) has twice the abundance of carbon relative to nitrogen, leading the researchers to think that something else delivered the volatiles.
To start, the team set out to recreate the high-temperature and high-pressure conditions of a primordial planetary body in the lab. They added volatiles to a mixture of silicates — representing the body’s crust and mantle — and iron-nickel alloy, representing the body’s core. Then the scientists watched to see how the volatiles evolved as the metal alloy separated from the silicates.
The scientists paid particular attention to carbon’s behavior: if it were drawn into the metal alloy, then this would result in a low carbon-to-nitrogen ratio in the silicates. If, however, carbon were drawn into the silicates, they would have a higher carbon-to-nitrogen ratio.
In the experiment carbon’s behavior depended on the amount of sulfur in the system, explains coauthor Rajdeep Dasgupta. With no sulfur present, most carbon was drawn into the metal alloy, but if the system contained 25% sulfur, most carbon floated to the silicate-rich portion, giving it the high carbon-to-nitrogen ratio found in Earth’s mantle.
Volatiles from Theia
With these results in hand, the team was ready to run simulations to see what kind of body had brought volatiles to Earth. Coauthor Chenguang Sun says that the team ran about a billion scenarios with impactors of different sizes and compositions, looking for ones that could explain present-day carbon, nitrogen, and sulfur abundances. The most likely culprit? A Mars-sized planetary body — just like what’s predicted in Moon formation scenarios.
In the simulations, this scenario works out if the volatile-rich body collided with Earth when our planet had about 90% of its current mass, says Grewal. Earth would have absorbed the small planet upon impact — the body’s core would have fused with Earth’s core, giving Earth’s center a boost in sulfur content. Meanwhile, the two mantles would have fused as well. While the merger would have diluted the total abundance of carbon and nitrogen, the carbon-to-nitrogen ratio would have stayed the same. As a result, Earth would have gained the remainder of its mass and the volatiles necessary for life. Grewal points out that the simulations of such an impact give the 40:1 ratio of carbon to nitrogen that’s seen in Earth’s silicate-based crust and mantle.
Rajdeep Dasgupta
But not all agree that the origin of Earth’s volatiles is settled. “The idea works better for carbon, nitrogen, and sulfur than it does for H2O, which probably has a more complex origin,” cautions Marc Hirschmann (University of Minnesota), who was not involved in the study. He adds that volatiles might not have been delivered in a single event: “There were lots of other opportunities for Earth to gain some of its volatiles both earlier and later.”
 6
6









Comments
Rod
January 31, 2019 at 11:11 am
Thanks for this good report. I note the proto-earth was about 90% present mass before the giant impact in this model. Other models I read about, the proto-earth is about 65% of present mass. Do we know the initial length of day before the giant impact and what the length of day was after the giant impact? Angular momentum has been an issue with the Earth-Moon system in simulations of the origin of the two as a pair.
You must be logged in to post a comment.
Spacivity
February 2, 2019 at 2:04 pm
My thoughts about the development of Earth are that at one point, the Earth was mostly formed about the same as Mars millions of years ago and it was still very hot. At that time, there was a comet also orbiting the sun. During that time, ice landed on Earth forming our atmosphere and continued to shed frozen matter onto Earth until it had shed the final core layer by bumping into Earth and adjusting our planet to its current orbit and the core of the comet remains orbiting us as our moon. While the atmosphere was forming, plants and animals (dinosaurs) began forming and living for years but when the final bump happened the dinosaurs were killed by the impact and the Earth started forming a cooler atmosphere which is what is current today. The global warming may be a result of the Earth's core heat working its way through the crust and out to the atmosphere and may be unstoppable, especially after the Arctic glaciers melt.
You must be logged in to post a comment.
Dobsonite
February 2, 2019 at 11:40 pm
Many decades ago, a study done in Human Circadian-Sleep cycles revealed that our biological clock is somewhere around 24.5 hours, not a straight 24 as was always assumed. As Mars rotation, I believe, is 24.5 earth hours plus or minus a tad, it certainly gives food for thought as to where and what "the spark" that kicked life off here may have come from.
All pure speculation, of course...but fun to think about.
You must be logged in to post a comment.
Roger Venable
February 3, 2019 at 8:08 am
This article doesn't solve the problem of the excess carbon, at all. You see, if Earth obtained its excess carbon from a planet-sized impactor, where did that planet-sized impactor get its excess carbon? Maybe the planet-sized impactor had an impact in its past -- an impact with another planet-sized impactor -- that conveyed the excess of carbon to it. And from whence did that second planet-sized impactor get its excess of carbon? And so on, ad infinitum. No answers at all.
If the planet-sized impactor can acquire an excess of carbon, presumably de novo without a presumption of a successiion of carbon-diluting impacts, with why can't Earth?
In order to solve the problem, what is needed is a more complete solar-system formation scenario in which some planets acquire a high carbon to nitrogen ratio as compared to the carbonaceous chondrites that we see today. To say that Earth's excess carbon came from such a planet doesn't explain a thing.
You must be logged in to post a comment.
Roger Venable
February 3, 2019 at 9:04 am
Oops. Earth is nitrogen rich, no carbon rich, so I got that part backward. So plug the word nitrogen into the forgoing comment, to replace the word carbon, and the reasoning will then be right.
You must be logged in to post a comment.
Rod
February 4, 2019 at 8:44 am
FYI. I did my own checks here. Using a proto-earth with 90% present mass of Earth, proto-moon with 90% present mass of Moon, initial orbital distance of 18395 miles in a circular orbit, the Moon's initial orbital period is 14.77 hours and angular size in the sky perhaps larger than 6.7 degrees. We have a number of free parameters in the computer models used to support the giant impact scenario for the origin of the Moon. These free parameters could create the Moon perhaps in the giant impact model but also bring destruction to Earth and the planet's habitability. The Earth-Moon system using the initial values for the proto-Earth and proto-Moon compared to present day measurements - shows just how much change in the Earth-Moon system is required to explain present day telescope observations of the pair when extrapolated over some 4.5 billion years.
You must be logged in to post a comment.
You must be logged in to post a comment.