NASA’s MAVEN mission has confirmed that the solar wind stripped the Red Planet of its atmosphere.

Lockheed Martin
Mars is the little planet that couldn’t. Its landscape, transformed by rivulets and lakes, has dried up. Its atmosphere is a wisp of what it once was. Based on the ratio of various elements’ isotopes — which aren’t currently what they should be if Mars always looked like this — planetary scientists suspect that the Red Planet has lost anywhere from 25% to 90% of its atmosphere over the last 4-ish billion years, with the estimates favoring at least 50%.
One popular explanation among planetary scientists has been that Mars’s atmosphere was stripped away by the solar wind, the barrage of charged particles that stream out from the Sun at 400 km/s (900,000 mph) and up. Now, scientists with NASA’s MAVEN orbiter mission have confirmed this hypothesis.
The MAVEN spacecraft loops around Mars, dipping in and out of its upper atmosphere. Using the craft’s measurements, Bruce Jakosky (University of Colorado, Boulder) and colleagues determined the levels of two isotopes of argon, argon-36 and the heavier argon-38. Argon is a noble gas (in the rightmost column of the Periodic Table of elements), which means it doesn’t interact chemically with the surface. The only way to get rid of it is to strip it from the atmosphere into space.
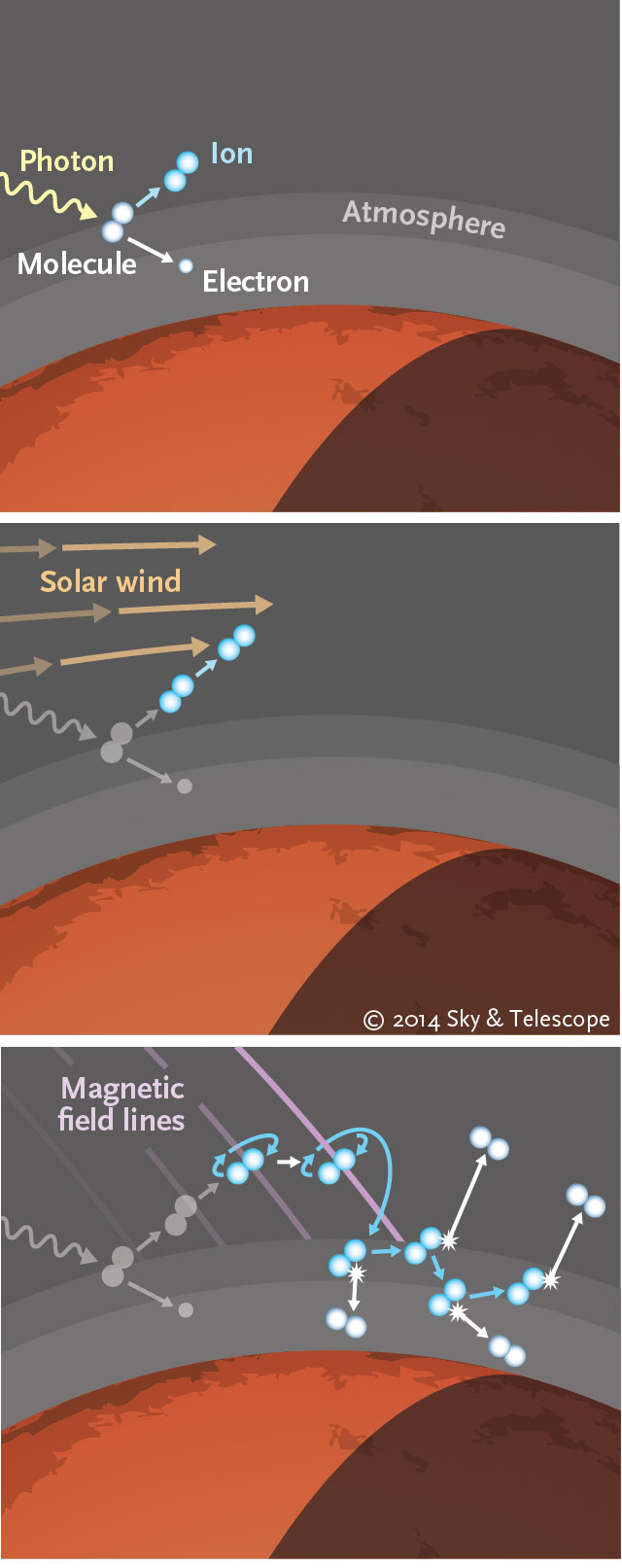
Casey Reed / Sky & Telescope. All Rights Reserved.
Argon’s heavier isotope naturally settles lower in the Martian air than the lighter one, creating a predictable ratio of argon-36 to argon-38 in the upper atmosphere. This differentiation leaves the lighter isotope more susceptible to being torn away by the solar wind in a process called sputtering.
In sputtering, the atmosphere colludes with the solar wind against the planet. Ultraviolet photons from the Sun first knock electrons from the atmosphere’s atoms and molecules, forming ions. The solar wind then picks up these ions, whirling them around and flinging some of them back into the atmosphere, where they collide with neutral atoms and molecules there — such as argon-36 —and “sputter” them every which way, including out of the atmosphere entirely.
MAVEN’s observations show that today’s Mars has far too little argon-36, if it started out with levels similar to Earth and other solar system objects. To explain current levels, the planet has lost roughly two-thirds of its atmospheric argon to sputtering over its history, the team concludes in the March 31st Science.
This result agrees with previous Mars studies, including a 2013 study by Sushil Atreya (University of Michigan) and others using Curiosity rover data. (Incidentally, the MAVEN result incorporates Curiosity’s measurements.) The percentage itself is not the news; rather, MAVEN’s notable contribution is the evidence for how the argon isotopes separate and how the argon is lost.
Although argon is a loner element, it wouldn’t have left alone. Other atmospheric constituents would have escaped Mars when it did. Jakosky’s team estimates that, based on the argon ratio, Mars has lost at least half a bar (where 1 bar is the atmospheric pressure at sea level on Earth) of its primary atmospheric molecule, carbon dioxide. That’s enough to at least partially explain the loss of the planet’s ancient warmer, wetter climate.
Thus, MAVEN confirms that modern Mars is a frozen desert world in part because the solar wind blew off its insulation.
References:
Bruce M. Jakosky et al. “Mars’ Atmospheric History Derived from Upper-atmosphere Measurements of 38Ar/36Ar.” Science. March 31, 2017.
Learn more about the MAVEN mission in our September 2014 cover story.
 3
3









Comments
Lindsay
March 31, 2017 at 4:52 pm
Great article, Camille!
You must be logged in to post a comment.
DHAndrews
April 1, 2017 at 10:01 am
I AM 72 years old! When I was in the 4th grade (1953) I did a major project on the Solar system. It was general knowledge at that time that Earth and Mars began their Solar life with equivalent atmospheres but solar winds and ambient planetary temperatures resulted in Mars losing most of her atmosphere.
So, what have we been learning since then?
You must be logged in to post a comment.
Camille M. CarlislePost Author
April 3, 2017 at 2:02 pm
Ha, at first I reacted to this result the same way! It's certainly true that we've thought for some time that the solar wind blew off Mars's atmosphere, although there was a large segment of planetary scientists who thought the atmosphere must have reacted with/been sequestered in the Martian surface (it isn't, we now know). More recently, we've begun amassing data confirming that that hypothesis is actually true -- it's one thing to suspect it, but another to have the isotope fractions chemically confirm the suspicion. The MAVEN result is a further confirmation; its contribution is not the percentage of atmosphere lost or that it was lost, but confirming the mechanism by which it happened.
You must be logged in to post a comment.
You must be logged in to post a comment.