Does a recent study solve the long-debated solar abundance problem?
An international team of astronomers says it has just solved a solar mystery that’s stood for well over a decade.
There are two ways to work out what the Sun is made of. The first, called spectroscopy, looks at the Sun’s spectrum. Precise wavelengths of light are missing from this spectrum because they’ve been swallowed by different elements within the Sun.
The second method is called helioseismology and measures sound waves bouncing around inside the Sun. The waves sometimes make it to the solar surface, causing it to pulse. The chemical makeup of the Sun alters how these sound waves travel, so their unique patterns are another way to take a solar inventory.
Using these tools to understand what the Sun is made of is important work, because we use the Sun as a benchmark for modelling all the other stars in the universe.
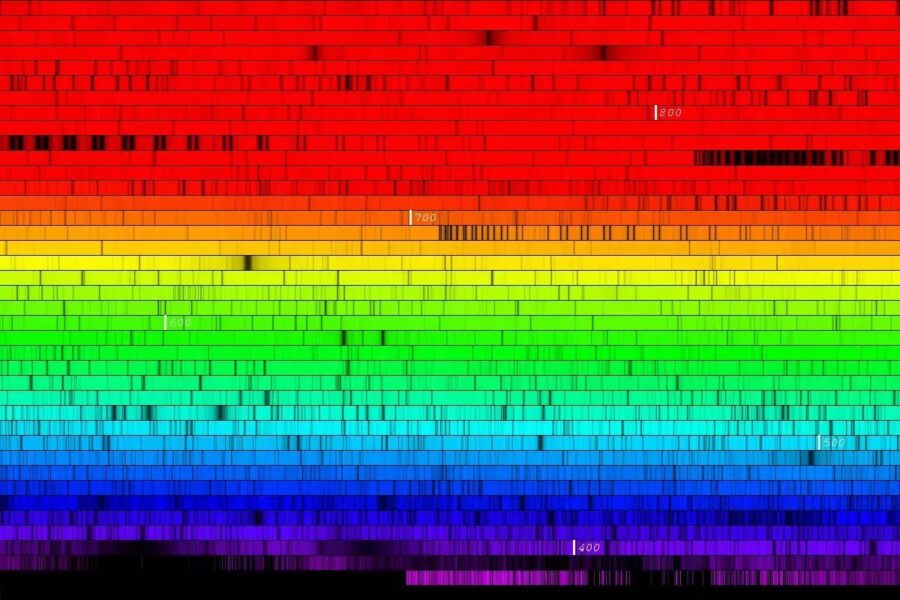
M. Bergemann / MPIA / NARVAL@TBL
Initially these two methods were in harmonious agreement. But in the early 2000s, as astronomers started using new and improved techniques, the measurements diverged. Specifically, they disagree on the amount of metals the Sun contains — that’s astrospeak for elements heavier than hydrogen and helium.
There have been numerous attempts to resolve this so-called solar abundance problem ever since, with everything from the solar wind to exotic physics being touted as an explanation (see S&T’s April 2021 cover story for details).
Now, it seems, there may be no abundance problem to solve after all. At least according to new research published in the May Astronomy & Astrophysics.
Ekaterina Magg (Max Planck Institute for Astronomy, Germany) and her colleagues have revisited the chemical abundances derived through spectroscopy. The necessary calculations are made easier if astronomers assume that the patch of the Sun they are looking at has a consistent temperature, something they refer to as local thermal equilibrium (LTE). However, they also know this is an inaccurate assumption.
By redoing the calculations to incorporate a more complex interaction between atoms and radiation, as well as the latest data, Magg and her collaborators are claiming a breakthrough.
“According to our analysis the Sun contains 26% more elements heavier than helium than previous studies had deduced,” Magg says. Using this new result produces a solar model that is consistent with all the information we have about the Sun’s present day structure, including what we know from sound waves, without the need for exotic physics.
In other words, the spectroscopists and helioseismologists appear reconciled again after two decades at loggerheads.
Or are they? “The solar abundance problem is far from resolved,” says Gaël Buldgen (University of Geneva, Switzerland).
Buldgen, a helioseismologist, cautions that Magg’s team compared their new spectroscopic models to a very limited model of the Sun, one that only uses a basic recipe for the Sun’s internal physics. “They completely neglect rotation inside the Sun, for example,” Buldgensays. “Once you include the effects of rotation, the agreement they find is broken.”
One of the elements involved in this debate is lithium. The lithium abundance predicted by standard solar models is much higher than what astronomers actually observe at the solar surface. Buldgen is a coauthor of a new paper out this week in Nature Astronomy, in which he and his collaborators argue that instabilities due to solar rotation and magnetic fields drag lithium — the lightest metal — away from the solar surface.
That, he says, is why astronomers see less lithium at the solar surface than they expected. This work suggests that rotation plays an important role in the chemical abundances we observe on the Sun, but Magg’s team haven’t yet built it into their work.
So, for now, it seems we remain at an impasse. More work is necessary before we can tell whether the solar abundance problem really has been solved, or whether yet another potential explanation will fall by the wayside.
 1
1








Comments
Jay Schufman
May 27, 2022 at 5:55 pm
Congratulations to Prof. Ekaterina Magg and her team on an elegant solution to a vexing inquiry! Bravo!
Regarding Dr. Buldgen rebuttal, it may be that the “magic number” nucleon stability of Be (4/9) is greater than Li (3/7).
You must be logged in to post a comment.
You must be logged in to post a comment.